 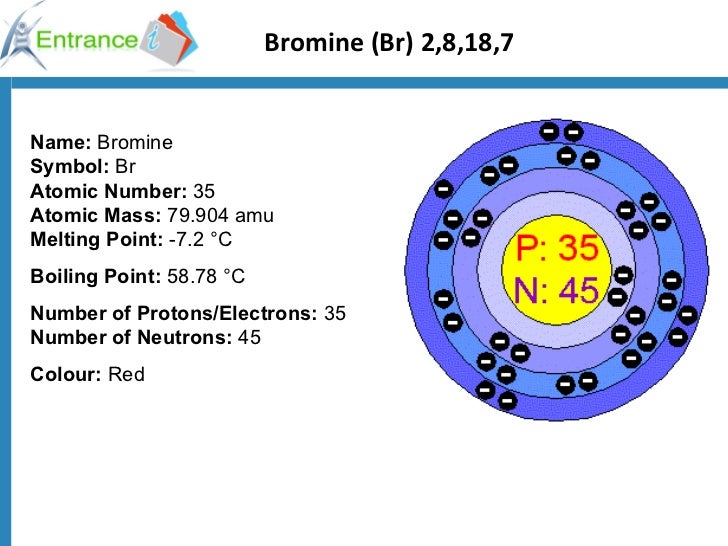
135Xe reactor poisoning played a major role in the Chernobyl disaster. As a result, the designers made provisions in the design to increase the reactor's reactivity (the number of neutrons per fission that go on to fission other atoms of nuclear fuel). This was discovered in the earliest nuclear reactors built by the Manhattan Project for plutonium production.  
The yield of 135Xe for uranium fission is 6.3% about 95% of 135Xe originates from decay of 135I.ġ35Xe is the most powerful known neutron absorber, with a cross section for thermal neutrons of 2.6×10 6 barns, so it acts as a " poison" that can slow or stop the chain reaction after a period of operation. 135I undergoes beta decay with half-life of 6.57 hours to 135Xe. It builds up in the reactor in the rate proportional to the rate of fission, which is proportional to the reactor thermal power. One of the common fission products is 135Te, which undergoes beta decay with half-life of 19 seconds to 135I. Buildup of stable or long-lived neutron poisons is called reactor slagging.įission products decay and burnup The buildup of short-lived fission products acting as nuclear poisons is called reactor poisoning, or xenon poisoning. The presence of 135I and 135Xe in the reactor is one of the main reasons for its power fluctuations in reaction to change of control rod positions. When 135Xe builds up in the fuel rods of a reactor, it significantly lowers their reactivity, by absorbing a significant amount of the neutrons that provide the nuclear reaction. 135I is a weak neutron absorber, while 135Xe is the strongest known neutron absorber. The main isotope responsible is 135Xe, mainly produced by natural decay of 135I. The iodine pit, also called the iodine hole or xenon pit, is a temporary disabling of a nuclear reactor due to buildup of short- lived nuclear poisons in the reactor core. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
